HEART DISEASES
Etiology and Symptoms
Correct diagnosis -
Application
of the most appropriate treatment!

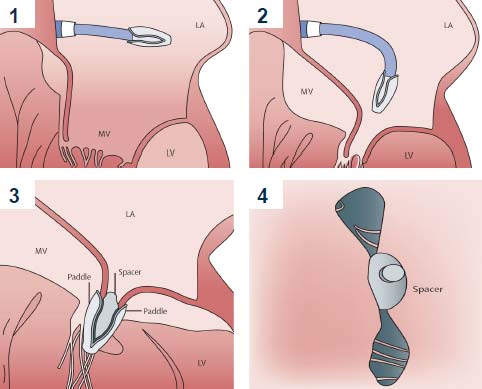
Mitral Valve Regurgitation
Is one of the most common heart valve disease occurring in almost 25% of the general population. It is estimated that almost the 7% of the general population above 75 years old have at least moderate degree of mitral regurgitation.
The heart has four cardiac valves: aortic, mitral, tricuspid and pulmonary valve. All of them are responsible for the correct direction of the blood flow.
Mitral valve regurgitation — also called mitral insufficiency is a condition in which your heart’s mitral valve doesn’t close tightly, allowing blood to flow backward in your heart. If the mitral valve regurgitation is significant, blood can’t move through your heart or to the rest of your body as efficiently, making you feel tired or out of breath.
Mitral regurgitation is due to damage to any of the individual parts of the valvular or sub-valvular mechanism (organic-degenerative) and / or to dilatation of the heart chambers (left ventricle +/- left atrium). In chronic situations the disease occurs progressively, while in acute is characterized by a sudden onset.
The Mitral regurgitation arises from various causes such as: rheumatic fever (nowadays it is rarely seen due to improved of the general health conditions), coronary heart disease, poor congenital structure of one and / or both mitral valve leaflets and, finally, due to dilation of the left ventricle causing mitral annulus dilation.
Sudden onset of mitral regurgitation is due to: myocardial infarction, infectious endocarditis, or injury.
The disease is characterized by shortness of breath initially only on exercise, fatigue and palpitations (extra systoles). Patients are prone to atrial fibrillation due to the large atrial dilatation. As a rule, we have of varying severity heart failure symptoms depending on the degree of insufficiency and the degree of adaptation of cardiac chambers (left ventricle) to volume overload situation. In acute insufficiency, acute pulmonary edema is possible.
Clinical and laboratory findings of chronic and acute mitral regurgitation | ||
| Chronic mitral regurgitation | Acute mitral regurgitation |
Symptoms |
|
|
Objective findings |
|
|
Laboratory and paraclinical tests |
|
|
Medical treatment improves and relieves temporarily the symptoms of mitral regurgitation. However, since the correction/repair problem is mainly mechanical, it does not cure it. It is a complex anatomical problem and this is the reason why the surgical approach i.e. the of our mitral valve or the implantation of a new artificial valve, does not always give positive results. Therapeutic intervention depends on the cause of the insufficiency and the general condition of the heart.
- In degenerative mitral regurgitation where the left ventricle is not involved in the pathology of the insufficiency, plastic valve correction (repair) or mechanical valve implantation is often effective.
- In acute mitral regurgitation, surgical treatment must be performed immediately in order to save the patient.
- In ischemic etiology due to partial or total dysfunction of the left ventricle due to active ischemia or old myocardial infarction, reperfusion surgery is of paramount importance. Sometimes the combination of reperfusion with surgical correction of the valve gives satisfactory results.
- In the case of functional mitral regurgitation due to advanced left ventricular dilatation and impaired cardiac output, the heart is suddenly in a high-pressure state immediately after correction of the insufficiency leading to collapse of the heart.
The surgical treatment is done with the help of an extracorporeal circulation machine that maintains perspiration and oxygenation during operation process. In the case of mitral valve replacement, the new valve prosthesis can be mechanical or biological. The latter does not require permanent anticoagulant treatment (Sintrom, Warfarin), but wears out faster (average lifespan of about 10 years) and is therefore preferred at older ages, where there is higher risk of bleeding. Therefore, the decision for surgical management must be individualized after a detailed weighing of several individual factors.
In the last decade, Transcutaneous Mitral Valve Repair has been successfully introduced into the therapeutic quiver, covering a large gap of patients, which are characterized as inoperable or of high surgical risk.
Coronary heart Disease
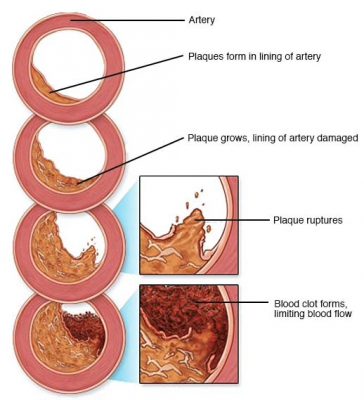
Coronary disease is one of the leading cause of death in modern societies, usually due to stenotic lesions of coronary arteries (arteries of the heart) due to atherosclerosis.
For descriptive, clinical and prognostic reasons, angina (chest pain) is classified into:
– Effort angina: when there is no substantial worsening of symptoms for several months. In the majority of patients, stable angina is due to the presence of “stable” atherosclerotic plaque in at least one large artery of the heart. Indicatively, angina symptoms appear when the stenosis is now approaching 75%.
– Unstable angina: usually due to ulceration or rupture of atherosclerotic coronary plaque and formation of an endovascular thrombus. Clinically the severity of the symptoms worsens abruptly without any obvious cause of increased myocardial oxygen demand or when angina occurs at rest.
– Prinzmetal angina: refers to angina attacks that develop without a clear trigger and are accompanied by ischemic changes (ST-segment elevation) on the electrocardiogram. It is most commonly due to a coronary artery spasm, and so is also called “vasoconstrictor”. This form of angina is more common on women.
The main clinical manifestation of coronary heart disease is characterized by angina. Angina is a clinical syndrome characterized by discomfort in the chest, lower jaw, shoulders, back or arms. In its typical form, angina is released during exercise or emotional loading and is relieved by the use of nitroglycerin (medication than cause vasodilatation).
However, angina often occurs in patients with no-coronary heart disease but in valvular heart disease, hypertrophic cardiomyopathy or poorly controlled hypertension. Angina may also occur in people with normal coronary arteries (Prinzmetal angina).
Factors that accelerate the progression of atherosclerotic disease (called cardiovascular risk factors) are: aging, gender, family history, hypertension, smoking, hyperlipidemia and obesity.
The treatment of coronary heart disease has two main goals: 1) prevention of future coronary events (such as: acute myocardial infarction, acute coronary syndromes and need for coronary reperfusion), and 2) the remission of symptoms (reduction in the frequency and severity of angina attacks) and improvement of quality of life.

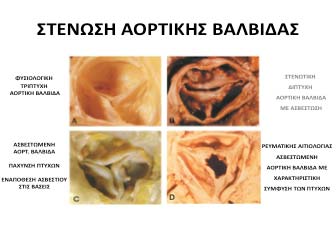
Aortic valve stenosis
The heart consists of four cardiac valves: the aortic, the mitral, the pulmonary and the tricuspid. Cardiac valves keep blood flowing in the correct direction. Each valve has flaps (cusps or leaflets) that open and close once during each heartbeat. Valvular disease is defined as the damage of at least one of the heart valves, constituting a major cause of morbidity and mortality.
The aortic valve opens when the heart (left ventricle) contracts, pushing blood to all organs in the body. Aortic valve stenosis is defined as the restriction of blood flow during contraction of the left ventricle at the level of the valve. It is the most common type of valvular heart disease in aging population characterized by progressive degeneration and thickening of the cusps of the aortic valve. When the aortic valve opening is narrowed, the heart must work harder to pump enough blood into the aorta and thus to the rest of your body. This result, initially, in hypertrophy and then gradually in fatigue and dilation of the heart chambers (left ventricle).
Narrowing of the valve can be due to a variety of causes, such as: after a rheumatic fever or as a result of progressive degeneration (aging). The increase in the population of elderly patients contributes to the increase in the incidence of aortic valve stenosis due to degeneration. The degenerative process starts from the base of the aortic cusps, causing a reduction of the orifice area, while stenosis occurs in the 6th-7th decade of our life. In cases of bicuspid aortic valve (congenital anomaly of the valve), stenosis occurs faster in the 4th-5th decade.
In a few cases the aortic stenosis is not exactly at the level of the valve (valvular), but sub-valvular or supra-valvular, due to the presence of a fibrous or muscular membrane at the level below or above the valve, respectively.
Factors that accelerate the progression of the disease are:
- Aging,
- Presence of coronary heart disease,
- Having cardiovascular risk factors, such as: diabetes, high cholesterol, smoking and high blood pressure (hypertension),
- Certain heart conditions present at birth (congenital heart disease) such as a bicuspid aortic valve,
- History of infections that can affect the heart
- Chronic kidney disease
- History of radiation therapy to the chest
Aortic valve stenosis ranges from mild to severe. Signs and symptoms generally occur when narrowing of the valve is severe. Some people with aortic valve stenosis may not have symptoms for many years. Indeed, the disease is characterized by a long asymptomatic period during which mortality is very low. When symptoms start, the disease is advanced and is characterized by a poor short-term prognosis.
The classic symptoms of aortic valve stenosis are:
- Chest pain or Angina
- Syncope (in 15% of patients which usually occurs during exercise).
- Symptoms of congestive heart failure such as: shortness of breath with activity, nocturnal dry cough, orthopnea, paroxysmal nocturnal dyspnea and hemoptysis.
- Palpitation feeling, rapid or fluttering heartbeat. If atrial fibrillation occurs, the lack of left atrial lactation worsens the condition and leads more quickly to heart failure.
The crucial issue is not the treatment itself, but the choice of the most appropriate time to apply it. From the moment the symptoms start, the disease accelerates; the anatomy of the heart and its function changes relative fast, and as a result the average prognosis in untreated patients is worse (2 – 5 years). Delayed treatment carries the risk of developing heart failure or sudden cardiac death. Therefore, recent data suggest an early management of the aortic stenosis and in general of all severe heart valvular diseases.
Medication relieves only the symptoms temporarily, but does not cure the mechanical problem of the stenosis. The only definitive treatment is the aortic valve replacement.
Until recently, surgery was the only treatment option in such patients with severe stenosis, with satisfactory immediate and long-term results. The surgical correction is done by implantation of a new prosthesis (mechanical or biological valve) and with the help of extracorporeal circulation machine that maintains perspiration and oxygenation during the operation. Mortality ranges between 1 – 4%.
However, when symptoms appear, many patients are already old and often with severe comorbidities. Therefore, such patients have a high or even prohibitive surgical risk and thus, such patients often remained uncured poor prognosis. The relative new method of transcutaneous replacement or implantation of the aortic valve (through catheters) revolutionized the field of valvular heart diseases and nowadays is the treatment of choice not only in high surgical / inoperable patients but even in patients who are considered as of intermediate-high surgical risk.

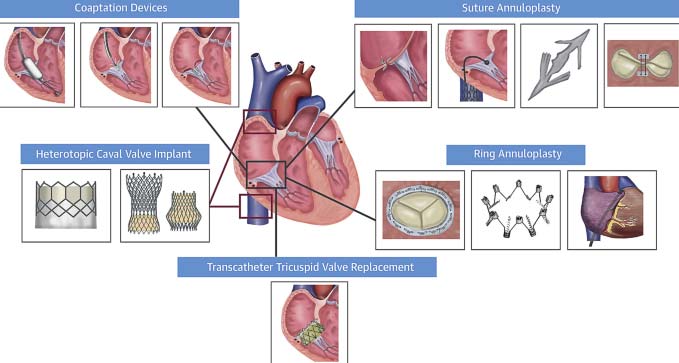
Tricuspid Valve Regurgitation
The heart consists of four cardiac valves: the aortic, the mitral, the pulmonary and the tricuspid. Cardiac valves keep blood flowing in the correct direction. Each valve has flaps (cusps or leaflets) that open and close once during each heartbeat. Valvular disease is defined as the damage of at least one of the heart valves, constituting a major cause of morbidity and mortality.
Regardless of the etiology, the Tricuspid regurgitation or insufficiency is characterized by reflux (return) of blood through the tricuspid orifice causing dilatation and dysfunction of the right chambers of the heart. We emphasize that a small degree (trivial or mild) of regurgitation is often seen in normal individuals.
Tricuspid valve regurgitation can be due to a variety of causes:
- Usually is functional or secondary due to structural anatomical changes of the valve.
- Rarely is organic or degenerative and may be due to: rheumatic fever, endocarditis, prolapse of the leaflets, tumors, connective tissue diseases, previous radiotherapy and congenital abnormalities such as Ebstein disease. Also, the use of anorexic drugs can cause a such regurgitation.
In most of the cases, the symptoms and findings of tricuspid insufficiency coexist with other manifestations of left heart failure due to mitral valve dysfunction. This is usually accompanied by shortness of breath.
The patient often complains of: fatigue on mild activity, weakness, swelling in the lower extremities, pain in the upper right abdomen and ascites.
In secondary Tricuspid regurgitation, correction of the underlying disease usually improves the degree of valve dysfunction, reducing the systolic pressure in the pulmonary artery. In addition, medication in cases of secondary pulmonary hypertension can significantly reduce the degree of tricuspid regurgitation.
There are indications for invasive treatment of the tricuspid valve in cases such as: acute tricuspid regurgitation, valve endocarditis and in cases of valve injury.
On the contrary, the treatment of chronic forms of valve insufficiency requires the consideration of many factors. The function of the right abdomen plays an important role. In neglected cases in which the function of the right abdomen is severely affected, invasive treatment of the tricuspid can further worsen the clinical picture of the patient.
Recently, the transcatheter approach has been introduced with high degree of safety and satisfactory results, covering a large gap of non-surgical patients or high surgical risk. In the next years we expect that this transcutaneous approach is going to expand.

Hypertrophic Cardiomyopathy (HCM)
Hypertrophic cardiomyopathy (HCM), affecting 1 to 500 people in general population, is a genetic disease in which the heart muscle becomes thickened (hypertrophied) combined with rearrangement of myocardial cells (myofiber disarray) and fibrosis. This makes it hard for the heart to relax and reduces the amount of blood the ventricle can hold and send to the body with each heartbeat. Hypertrophic cardiomyopathy often goes undiagnosed because many people with the disease have few, if any, symptoms. However, in a small number of people with HCM, the thickened heart muscle can cause shortness of breath, chest pain or changes in the heart’s electrical system, resulting in life-threatening irregular heart rhythms (arrhythmias) or sudden death.
Hypertrophic cardiomyopathy is usually caused by changes in genes (gene mutations) that cause the heart muscle to thicken.
- In 60% of the cases the disease is due to mutations of genes that encode sarcomeres.
- In 5-10% of the cases the disease is due to metabolic disorders such as: Danon, Anderson – Fabry, or ATPKinase – PRKAG2 disease and neuromuscular disorders such as Friedreich.
- In 25-30% of the cases the HCM is of unknowη
Signs and symptoms of hypertrophic cardiomyopathy might include one or more of the following:
- Chest pain, especially during exercise
- Fainting, especially during or just after exercise or exertion
- Heart murmur, which a health care provider might detect while listening to the heart
- Sensation of fast, fluttering or pounding heartbeats (palpitations)
- Shortness of breath, especially during exercise
Complications of hypertrophic cardiomyopathy can include:
- Atrial fibrillation.A thickened heart muscle and changes in the structure of heart cells can cause changes in the heart’s electrical system, resulting in fast or irregular heartbeats. Atrial fibrillation can also increase the risk of developing blood clots, which can travel to the brain and cause a stroke.
- Blocked blood flow.In many people, the thickened heart muscle blocks the blood flow leaving the heart, causing shortness of breath with exertion, chest pain, dizziness and fainting spells.
- Mitral valve disease.If the thickened heart muscle blocks the blood flow leaving the heart, the valve between the left atrium and left ventricle (mitral valve) might not close properly. As a result, blood can leak backward into the left atrium (mitral valve regurgitation), possibly making symptoms worse.
- Fainting (syncope).An irregular heartbeat or blockage of blood flow can sometimes cause fainting. Unexplained fainting can be related to sudden cardiac death, especially if it’s happened recently and in a young person.
- Sudden cardiac death.Rarely, hypertrophic cardiomyopathy can cause heart-related sudden death in people of all ages. Because many people with hypertrophic cardiomyopathy don’t realize they have it, sudden cardiac death might be the first sign of the condition. It can happen in seemingly healthy young people, including high school athletes and other young, active adults.
Your provider will likely order tests to diagnose hypertrophic cardiomyopathy (HCM) or rule out other conditions that can cause similar symptoms.
- Xray or Low dose Computer Tomography of Thorax
- Electrocardiogram (ECG).Sensors (electrodes) attached to adhesive pads are placed on the chest and sometimes the legs to measure electrical signals from the heart. An ECG can show irregular heart rhythms and signs of heart thickening.
25-48 hours Rhythm monitoring (Holter monitor). Your health care provider may recommend monitoring your heartbeat at home. A portable ECG device can be worn for a day or more to record the heart’s activity during daily activities. Threaten arrythmias can be diagnosed.
- An echocardiogram is commonly used to diagnose hypertrophic cardiomyopathy. This test uses sound waves (ultrasound) to see if the heart’s muscle is unusually thick. It also shows how well the heart’s chambers and valves are pumping blood.
- Cardiac Catheterization.
- Cardiac MRI.This test uses powerful magnets and radio waves to create images of the heart. It provides information about the heart muscle and how the heart and heart valves work. This test is often done with an echocardiogram.
- Stress test.A stress test often involves walking on a treadmill or riding a stationary bike while the heart is monitored. Exercise stress tests help reveal how the heart responds to physical activity.
- Genetic control.
The goals of hypertrophic cardiomyopathy treatment are to relieve symptoms and prevent complications and mainly sudden cardiac death in people at high risk. Treatment depends on the severity of symptoms. If you have cardiomyopathy and are pregnant or thinking about pregnancy, your health care provider might recommend that you see a doctor experienced in caring for women with high-risk pregnancies (perinatologist or maternal-fetal medicine specialist).
Medications can help reduce how strongly the heart muscle squeezes and slow the heart rate so that the heart can pump blood better. Medications to treat hypertrophic cardiomyopathy and its symptoms might include: Beta blockers such as metoprolol or propranolol. In case of Beta blocker contraindication calcium channel blockers such as verapamil or diltiazem. Heart rhythm drugs such as amiodarone. Finally, Blood thinners such as warfarin or NOACS to prevent blood clots if you have atrial fibrillation or the apical type of hypertrophic cardiomyopathy, which can increase the risk of sudden cardiac death.
- Κολποκοιλιακή βηματοδότηση Πρόσφατα δημοσιεύτηκε η μεγάλη πολυκεντρική μελέτη Μ-ΡΑΤΗΥ, που καταλήγει ότι οι ασθενείς που ωφελούνται περισσότερο είναι οι ηλικιωμένοι
- Septal ablation.This procedure destroys the thickened heart muscle with alcohol. The alcohol is injected through a long, thin tube (catheter) into the artery supplying blood to that area. Possible complications include disruption of the heart’s electrical system (heart block), which requires implantation of a pacemaker.
- Septal myectomy.This open-heart surgery might be recommended if medications don’t improve symptoms. It involves removing part of the thickened, overgrown wall (septum) between the heart chambers. Septal myectomy helps improve blood flow out of the heart and reduces backward flow of blood through the mitral valve (mitral regurgitation). The surgery can be done using different approaches, depending on the location of the thickened heart muscle. In one type, called apical myectomy, surgeons remove thickened heart muscle from near the tip of the heart. Sometimes the mitral valve is repaired at the same time.
Sudden death risk evaluation. Risk factors include:
- Successfully treated episode of sudden death
- Persistent and non-Persistent episodes of ventricular tachycardia
- Recurrent syncope episodes
- Drop of blood pressure during exercise
- Family history of sudden cardiac death
- Severe heart muscle thickening (30mm)
- Young patients (< 40 years old)
In such cases the implantation of an\ Implantable cardioverter-defibrillator (ICD) is the treatment of choice. An ICD is a small device that continuously monitors the heartbeat. It’s implanted in the chest like a pacemaker. If a life-threatening arrhythmia occurs, the ICD delivers precisely calibrated electrical shocks to restore the heart rhythm. Use of an ICD has been shown to help prevent sudden cardiac death, which occurs in a small number of people with hypertrophic cardiomyopathy.
Acute Coronary Syndrome (ACS) – Acute Myocardial Infarction
Cardiovascular disease is the leading cause of death (49%) in modern societies of western world. This fact is due to aging of population and bad nutrition habits, in addition to traditional cardiovascular risk factors such as: obesity, lack of exercise, smoking, high cholesterol levels, high blood pressure and diabetes mellitus.
The coronary artery disease is one of the leading causes of death in modern societies owing in stenotic lesions of coronary arteries in most cases due to atherosclerosis. Clinically is classified into: silent ischemia, stable angina, unstable angina, acute myocardial infarction and sudden cardiac death.


Although, all cardiological societies give a huge effort for the prevention of future coronary events and treatment of coronary artery disease and mainly the acute coronary syndrome, however, myocardial infarction remains one of the most important causes of mortality in western societies.
The Acute myocardial infarction (STEMI), most commonly, is caused by an acute occlusion of one of the epicardial coronary arteries secondary to acute atherosclerotic plaque rupture and thrombosis.
Medical history and symptom description are important for the correct diagnosis. The resting 12-lead electrocardiogram (ECG) is the first-line diagnostic tool in the assessment of patients with suspected acute coronary syndrome (ACS) and it is recommended to perform it within 10 min of the patient’s arrival in the emergency room.
Based on the electrocardiogram two categories of clinically syndromes should be differentiated:
This condition is termed ST-segment elevation ACS and generally reflects an acute total or subtotal coronary occlusion. Most patients will ultimately develop ST-segment elevation myocardial infarction (STEMI). The mainstay of treatment in these patients is immediate reperfusion by primary percutaneous coronary intervention.
(primary coronary angioplasty).
Patients with acute chest discomfort but no persistent ST-segment elevation exhibit ECG changes that may include: transient ST-segment elevation, persistent or transient ST-segment depression, T-wave inversion, flat T waves, or pseudo-normalization of T waves; or the ECG may be normal.
.
Pain relief and control of myocardial necrosis markers are a primary goal. Based on the mobilization of myocardial enzymes into the bloodstream, patients are classified into two categories: patients with acute coronary syndrome without ST-elevation when troponin is high, and patients with unstable angina when there is no increase in troponin in the blood.
The early and accurate diagnosis is of paramount importance. The mainstay of treatment in these patients is the immediate reperfusion therapy by primary percutaneous coronary intervention and therefore patients should be transferred to a PCI-capable facility as soon as possible.
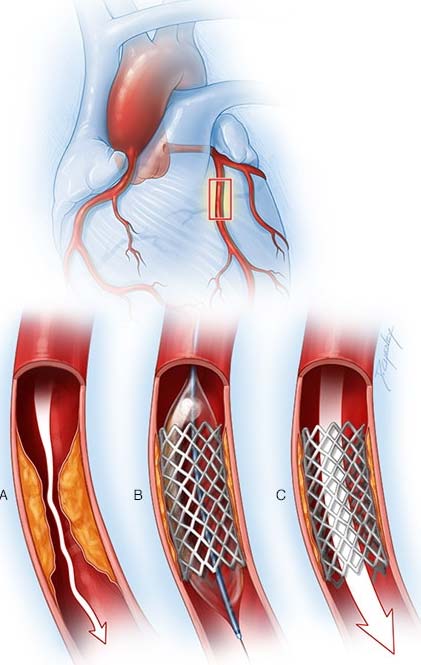
In cases where primary PCI is not an available option in a timely manner then fibrinolytic therapy is the choice of treatment. In case of thrombolysis failure, the patient should be transported immediately to a hospital with a hemodynamic laboratory, in order to perform angioplasty, which is superior to medication.
The primary coronary angioplasty (PCI) is a procedure used to immediately open the clogged heart culprit arteries. Angioplasty is combined with the placement of a small wire mesh tube called stent. The stent helps prop the artery open, decreasing its chance of narrowing again. Stent implantation in combination with proper blood-thinning medications ( antiplatelet medicines) help to reduce the risk of blood clots forming around the stent and thus closing again.
In conclusion primary coronary angioplasty with stent implantation and immediate/early coronary revascularization is the cornerstone of the treatment of the acute myocardial infarction since it improves prognosis, reducing mortality. Therefore, the early, accurate diagnosis and transportation of the patients to a PCI-capable facility as soon as possible is of crucial importance in cases of acute myocardial infarction.